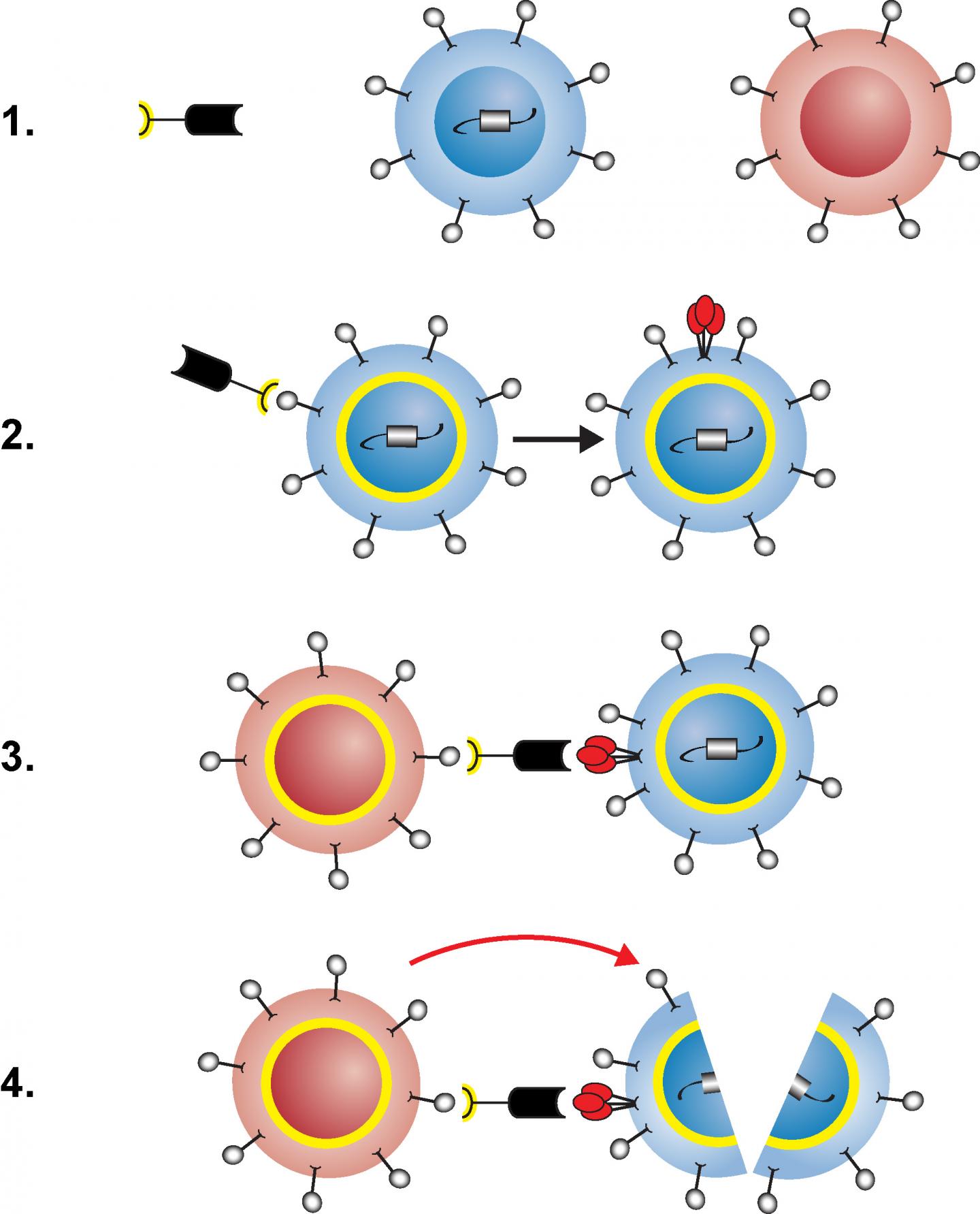
This is an illustration of how the engineered protein facilitates destruction of latently HIV-infected immune cells. Scientists at the National Institutes of Health (NIH) have created a protein that awakens resting immune cells infected with HIV and facilitates their destruction in laboratory studies. The protein potentially could contribute to a cure for HIV infection by helping deplete the reservoir of long-lived, latently HIV-infected cells that can start making the virus when a person stops taking anti-HIV drugs. Further studies in animals and people are needed to determine the viability of this approach.
The researchers found that the protein, called VRC07-αCD3, triggered the activation and killing of latently HIV-infected helper T cells when the cells were taken from patients on antiretroviral therapy and then incubated in the lab with the patients' own killer T cells. In addition, the scientists found a monkey-adapted version of the protein to be safe and well-tolerated when given to monkeys infected with a simian form of HIV and receiving antiretroviral therapy. The researchers are now studying the effectiveness of monkey-adapted VRC07-αCD3 in the animals.
The engineered protein has two ends: one activates T cells by binding to a surface molecule called the CD3 receptor, and the other--based on an antibody called VRC07--powerfully binds to more than 90 percent of HIV strains. VRC07-αCD3 facilitates the killing of latently HIV-infected cells in three steps. First, the CD3-binding end attaches to a resting, HIV-infected helper T cell, activating the cell so it starts making HIV and displaying pieces of virus on its surface. Next, the HIV-binding end of the protein latches onto those pieces of virus while the CD3-binding end attaches to a killer T cell, activating it and bringing it close to the helper T cell. Finally, the activated killer T cell destroys the HIV-infected helper T cell.
A team of scientists at the Vaccine Research Center (VRC) of the National Institute of Allergy and Infectious Diseases, part of NIH, created VRC07-αCD3 under the leadership of VRC Director John R. Mascola, M.D.; former VRC Director Gary J. Nabel, M.D., Ph.D.; and Richard A. Koup, M.D., VRC deputy director and chief of its immunology laboratory.
Source: NIH/National Institute of Allergy and Infectious Diseases
 Print Article
Print Article Mail to a Friend
Mail to a Friend
